Sulfuric acid is the largest-volume industrial chemical produced in the world (200 million tons per year). Concentrated sulfuric acid (93-98 %) is used in the manufacture of fertilizers, explosives, dyes, and petroleum products.
The starting material for sulfuric acid manufacture is clean, dry sulfur dioxide (SO2) gas. This can be obtained by burning molten sulfur, from metallurgical off-gases or by decomposing spent sulfuric acid.
Over the last decades the contact process has been used to produce sulfuric acid, replacing the traditional «Lead Chamber» process dating back to the 18th Century.
In the contact process SO2 is oxidized to sulfur trioxide (SO3) at high temperature (about 450°C) in the presence of a vanadium catalyst. SO3 then is dissolved in concentrated sulfuric acid forming fuming sulfuric acid (oleum). This can then be reacted safely with water to produce concentrated sulfuric acid.
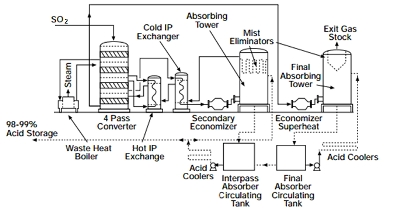
Major changes have been made in process and plant design to maximize energy recovery and then use this heat to generate high-pressure steam and/or electricity. The older «single absorption» process has been largely replaced by the «double absorption» process which increases yield of acid and reduces emissions. A typical flowchart for a double absorption sulfuric acid plant is shown in figure 1.