Phosphoric acid (H3PO4) can be produced by 3 main commercial methods: wet process, thermal process and dry kiln process. Wet process is by far the most common route and the acid can be used in phosphate fertilizers production (DAP, MAP, SPA). Thermal process phosphoric acid is of a much higher purity and is used in the manufacture of high grade chemicals, pharmaceuticals, detergents, food products, and other nonfertilizer products. The last method, using a rotary kiln, is a promising alternative because of its reduced environmental footprint and potential cost saving.
The concentration of phosphoric acid is normally expressed as % P2O5 (percent phosphoricanhydride) rather than % H3PO4 (percent phosphoric acid).
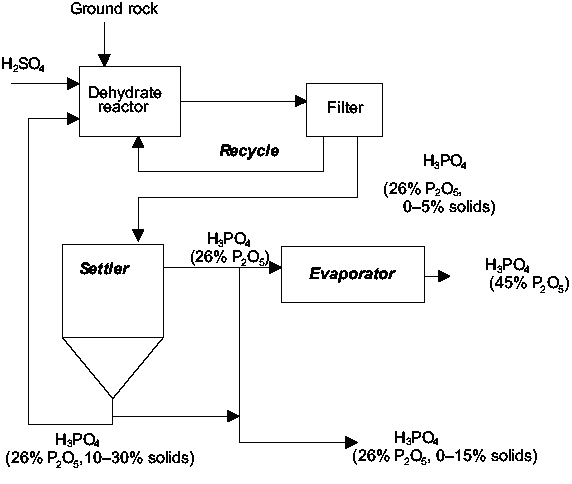
In a wet process facility (see figure 1), phosphoric acid is produced by reacting sulfuric acid (H2SO4) with naturally occurring phosphate rock. The reaction also forms calcium sulfate (CaSO4), commonly referred to as gypsum. Insoluble gypsum is separated from the reaction solution by filtration.
Operating conditions are generally selected so that the calcium sulfate will be precipitated in either the dihydrate or the hemihydrate form, thus producing 26-32% P2O5 at 70-80°C for dihydrate precipitation and 40-52% P2O5 at 90-110°C for hemihydrate precipitation. Further evaporation of the solvent can be performed for a high-concentration phosphoric acid.