Polycarbonate (PC) is an engineering plastic with outstanding transparency, impact resistance, and heat resistance. These unique properties have resulted in applications such as bulletproof windows, unbreakable lenses, compact discs, etc. About 2.7 million tons of polycarbonate are produced annually worldwide.
Manufacturing process of Polycarbonate
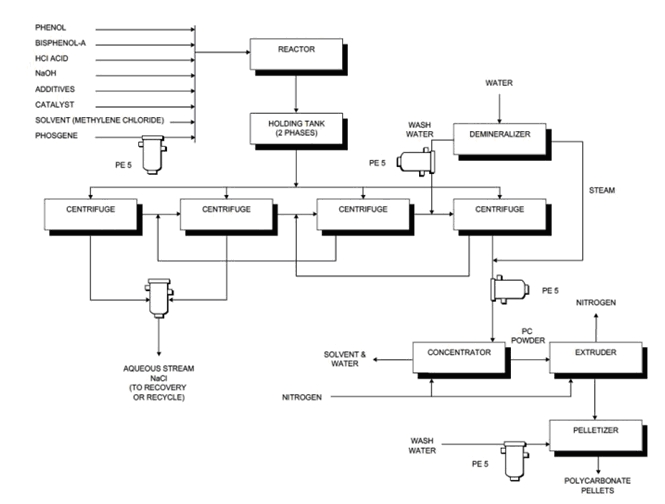
The most common manufacturing process is based on the reaction of bisphenol A (BPA or Bis-A) and phosgene in the interfacial polymerization process. Here, disodium saltof BPA dissolved in water reacts with phosgene dissolved in a chlorinated organic solvent such as CH2Cl2 (methylene chloride). However, the phosgene process entails a number of drawbacks including the toxicity of phosgene, the use of low-boiling-point solvent, and the large quantity of waste water containing methylene chloride which must be treated. The use of concentrated sodium hydroxide and hydrogen chloride adds the problem of corrosion that should be considered.
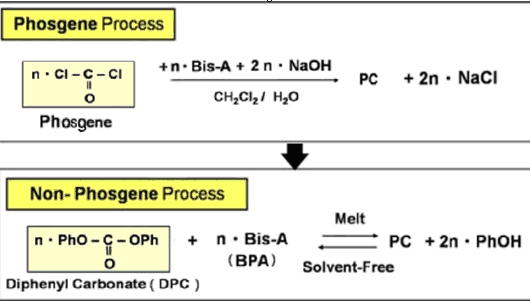
Nowadays, the production and use of phosgene in the factories have been very severely restricted worldwide. There are aSabic Innovative Plastics (formerly GE Plastics), Bayer, and Asahi/Chi Mei have independently developed and are using non-phosgene processes. They all take the same overall approach where polymerization relies on the transesterification of diphenyl carbonate (DPC) with bisphenol A. This is more commonly termed as the melt process which has the advantage of making a product undiluted form that may be pelletized directly. Disadvantages include the need for equipment to withstand high temperatures and high vacuum.
With lower plant construction costs and lower feedstock costs, it is anticipated that this kind of non-phosgene process will be widely adopted for PC production throughout the world.